

A research team from the School of Pharmacy, Tongji Medical College of Huazhong University of Science and Technology (HUST) has elucidated the biosynthetic pathway of emestrin A, a natural product with potent antitumor activity. The study, appearing in ACS Catalysis, identified a single bifunctional P450 enzyme that sequentially catalyzes two distinct reactions to construct the compound’s characteristic 15-membered macrocycle, offering new tools for natural product engineering.
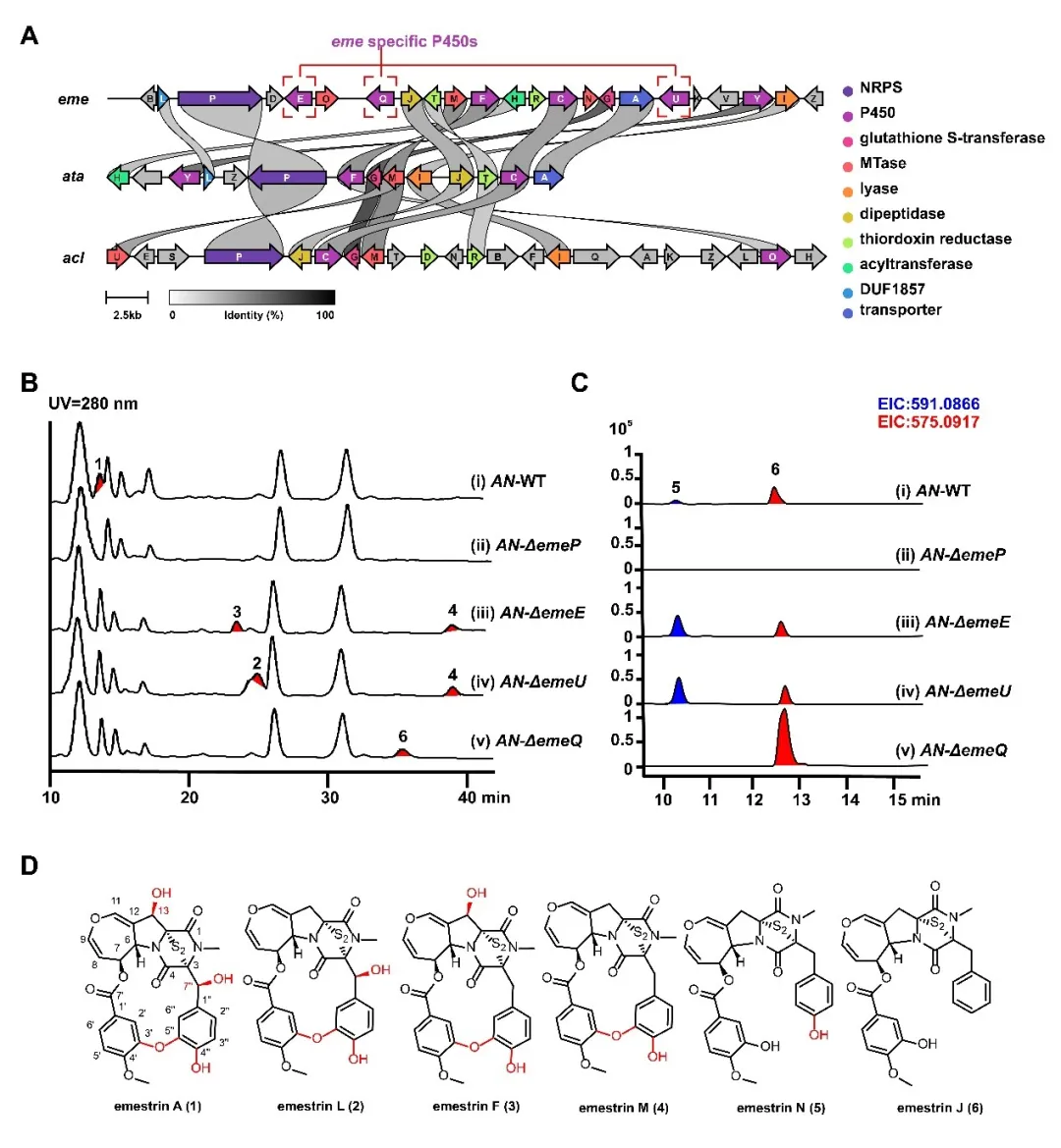
Emestrin A belongs to the epipolythiodioxopiperazine (ETP) family of natural products and has been a matter of active investigations for its unique ether-lactone macrocycle and strong antitumor activity. However, the formation of this macrocycle and the role of oxidative modifications in its biological activity had remained unclear.
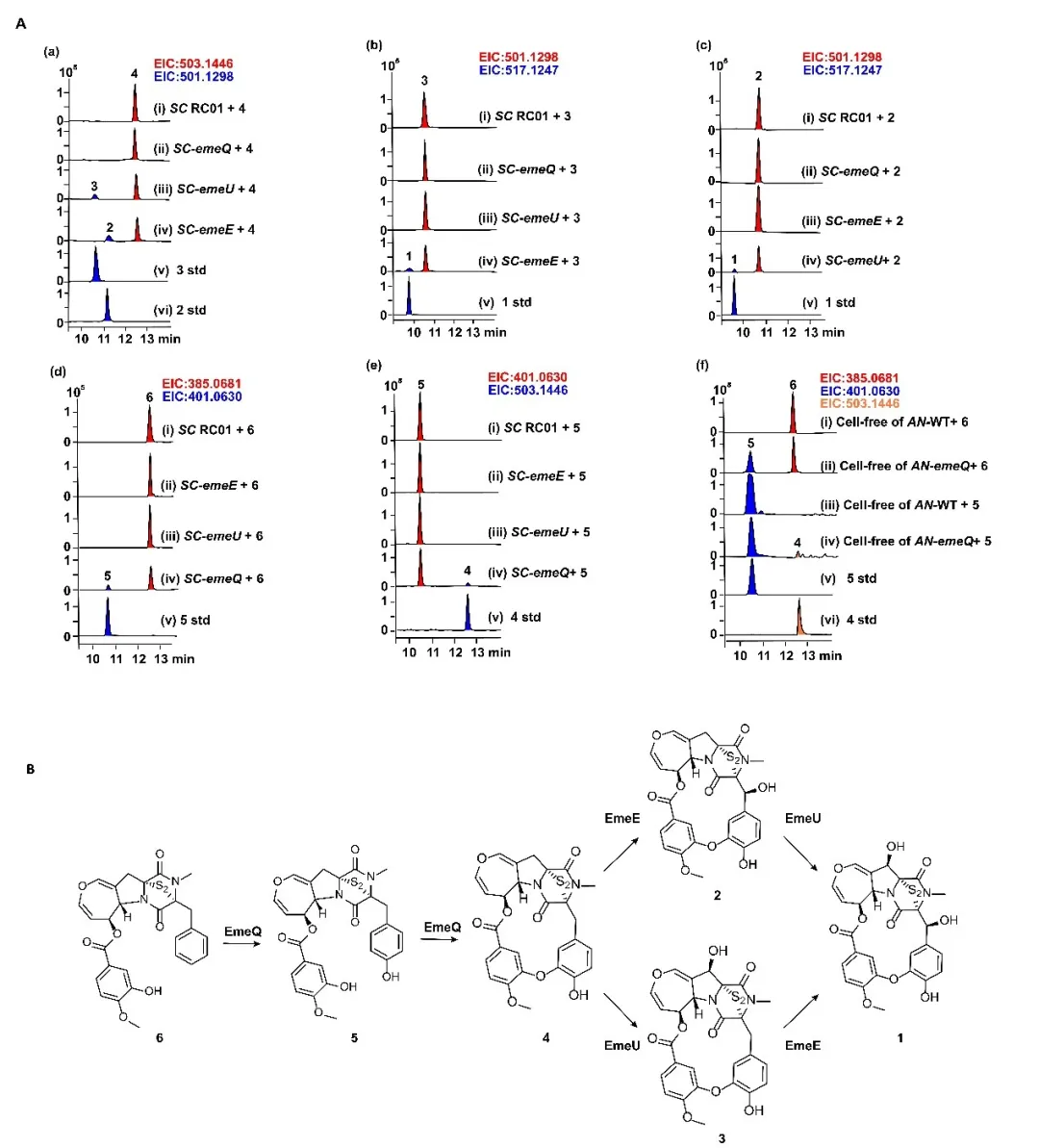
By integrating gene knockout, heterologous expression, feeding experiments, in vitro enzymatic assays, and computational simulations, the team systematically mapped out the late-stage biosynthetic pathway. They identified a gene cluster responsible for emestrin production and characterized the functional division of three P450 enzymes. Notably, one enzyme—EmeQ—was found to catalyze both aromatic hydroxylation and phenolic oxidative coupling, directly forming the macrocyclic structure in a single enzymatic cascade.
The researchers further elucidated the architectural basis of this dual functionality through molecular docking and site-directed mutagenesis. By mutating key residues in the enzyme’s active site, they successfully reprogrammed EmeQ into two highly efficient monofunctional variants, one dedicated to hydroxylation and the other to cyclization, thereby demonstrating the remarkable plasticity of fungal P450 enzymes.
The study also assessed the antitumor activity of various emestrin analogs, revealing that both the macrocyclic structure and the presence of specific free hydroxyl groups are critical for their potent activity.
This work provides new insights into the biosynthesis of complex natural products and offers valuable enzymatic tools for synthetic biology and the structural diversification of pharmacologically-active compounds.
Prof. ZHANG Yonghui, Associate Professor YE Ying, and Associate Professor LIU Junjun from the School of Pharmacy are the corresponding authors. PhD candidates YU Jingwen, LAI Suitian, and Associate Professor Li Qin are co-first authors. The research was supported by the National Key R&D Program of China and the National Natural Science Foundation of China.
