On October 26, the research group of Professor YUAN Shuiqiao of the Institute of Reproductive Health published an online paper entitled“hnRNPU in Sertoli cells cooperates with WT1 and is essential for testicular development by modulating transcriptional factorsSox8/9”.
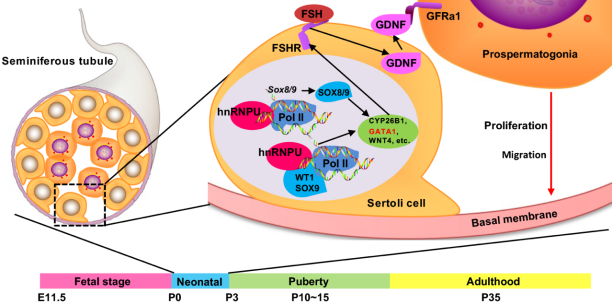
This paper reveals for the first time the important role of RNA-binding protein hnRNPU in Sertoli cells in male infertility, and finds that hnRNPU in mammalian Sertoli cells directly regulates the expression of important markers Sox8 and Sox9 in Sertoli C cells to regulate the target genes related to testicular development. O as to maintain the spermatogenic microenvironment of the testicular seminiferous tubules and the normal development and function of spermatogenic cells.

Up to 15% of couples worldwide suffer from infertility, of which about 50% are caused by male factors.Male fertility mainly depends on the continuous production of spermatozoa. Testicular seminiferous tubules are the most important site of spermatogenesis. Sertoli cells are the most important somatic cells in testicular seminiferous tubules, which provide a stable spermatogenic microenvironment for the development and differentiation of germ cells.Sertoli cells can maintain the shape of seminiferous tubules, provide nutrients and physical barrier protection for germ cells in seminiferous tubules, and play an indispensable role in the proliferation and migration of germ cells.It has been reported that the deletion of some proteins in Sertoli cells can lead to infertility. However, the molecular mechanism and regulatory network of how Sertoli cells regulate the development of germ cells and testis are still unclear.

On the basis of previous studies, Prof. YUAN's team found that RNA-binding protein hnRNPU was expressed in human and mouse Sertoli cells, and there was a high degree of conservation.Through Cre/LoxP technology, a conditional knockout mouse model of Hnrnpu gene in Sertoli cells was constructed for the first time. Phenotypic analysis showed that male mice lacking hnRNPU in Sertoli cells were completely infertile, their testes were significantly smaller, which could not produce sperm.The testis seminiferous tubules of neonatal knockout mice have been severely damaged, the structure of Sertoli cells is abnormal, and the germ cells can not proliferate and migrate normally, which ultimately leads to the failure of sperm production.
These findings suggest that hnRNPU protein in Sertoli cells plays an important role in the regulation of male fertility. In terms of molecular mechanism, hnRNPU can interact with Sertoli cell markers WT1 and SOX9 to form protein complexes, and the transcription levels of target genes of WT1 and SOX9 are changed after hnRNPU deletion.At the same time, our team found that two transcription factors Sox8/9, which are very important for testicular development, were down-regulated in Sertoli cells of Hnrnpu conditional knockout mice, and ChIP-qPCR was used to prove that hnRNPU could directly bind to the promoter region of Sox8/9 transcription factor. These results suggest that hnRNPU protein in Sertoli cells can bind to the promoter region of Sox8/9 and affect their transcriptional activity, and then regulate its downstream important target genes, ultimately affecting testicular development.The results of this study are expected to further improve the molecular regulation mechanism of male infertility, and provide a new theoretical basis for the diagnosis and treatment of non-obstructive azoospermia.
This achievement is an important breakthrough in spermatogenesis and male infertility regulation after Prof. YUAN Shuiqiao's research team explained the morbidity mechanism of male infertility in Development and PNas respectively in the first half of this year.
Paper linkage:https://www.thno.org/v11p10030.htm
