Metabolite-loaded extracellular vesicles reprogram macrophage metabolism to suppress tumor growth
WUHAN – A research team led by Prof. TANG Ke and HUANG Bo at the School of Basic Medicine, Tongji Medical College of Huazhong University of Science and Technology (HUST), has developed an innovative extracellular vesicle-based delivery system that reprograms tumor-associated macrophages using succinate, offering a promising new approach for cancer immunotherapy. The study was published on April 9 in Science Translational Medicine, a journal of the American Association for the Advancement of Science (AAAS).

The research builds on Prof. HUANG’s pioneering work since 2009 using tumor cell-derived extracellular vesicles as natural nanocarriers for drug delivery. In this latest study, the team used electroporation to efficiently load the metabolite succinate into extracellular vesicles, creating stable succinate-loaded micro particles (SMPs). When administered into multiple mouse tumor models—including hepatocellular carcinoma, melanoma, lung cancer, and breast cancer—SMPs effectively promoted the polarization of pro-tumor M2 macrophages into antitumor M1 macrophages, significantly inhibiting tumor growth. These findings were further validated using clinical tumor samples.
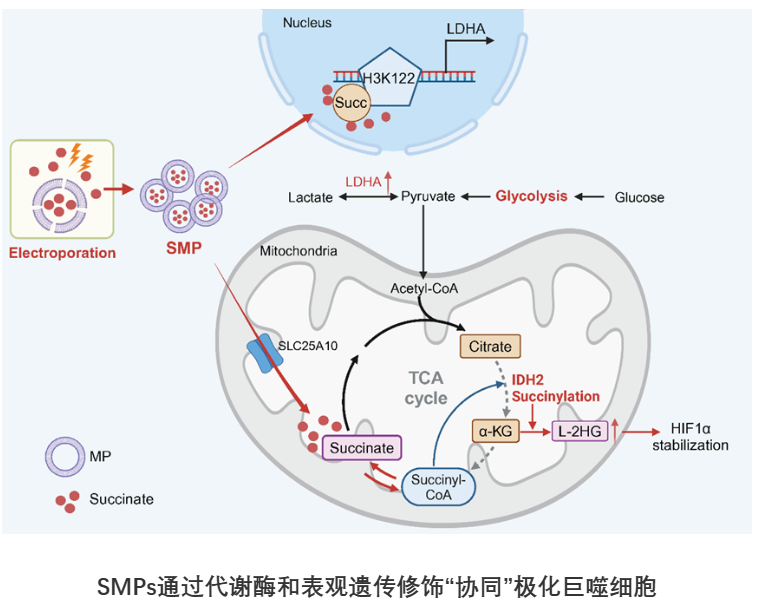
Mechanistic studies revealed that SMPs operate through a dual pathway: upon cellular entry, succinate is transported into mitochondria via SLC25A10, where it inhibits IDH2 activity and alters metabolic flux, leading to HIF-1α stabilization and enhanced glycolysis. Simultaneously, nuclear succinate modifies histone H3K122, promoting transcription of the glycolytic enzyme LDHA and reinforcing M1 macrophage polarization.
Unlike synthetic nanocarriers, SMPs are derived from natural cell membranes, possessing improved biocompatibility and reduced risk of toxicity. Since 2012, extracellular vesicle-based therapies developed by the team have entered clinical trials in China and were incorporated into the National Medical Service Technical Specifications in 2023.
The study provides a compelling cross-disciplinary strategy that combines vesicle delivery and metabolic reprogramming for cancer immunotherapy. The team continues to explore the broader applications of vesicle-based drug delivery systems for targeted therapy and immunomodulation.
Doctoral candidates LU Shuya and LI Jiaxiao are co-first authors of the paper. The research was supported by the National Key R&D Program, the National Natural Science Foundation of China, and the Chinese Academy of Medical Sciences Innovation Fund.
Article link:DOI: 10.1126/scitranslmed.adr4458
